“High-pressure behavior and crystal–fluid interaction under extreme conditions in paulingite [PAU-topology]”
- Authors
G.D. Gatta, K.S. Scheidl, T. Pippinger, R. Skála, Y. Lee, R. Miletich
- Journal
Microporous and Mesoporous Materials
vol.206, pp.34-41, 2015.04 - DOI
Abstract
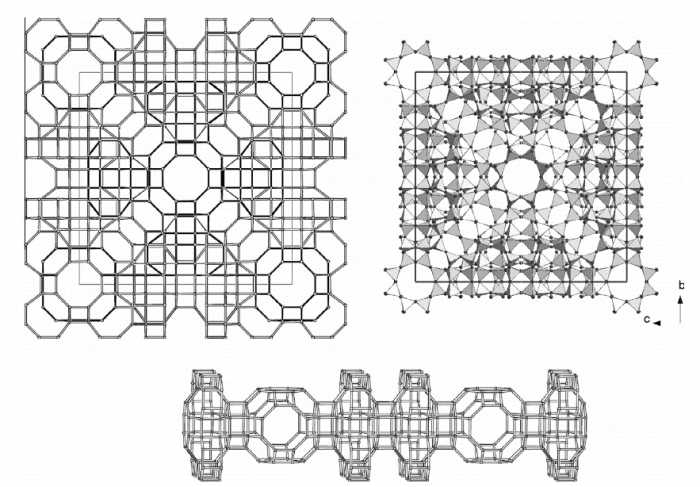
The compressional behavior and the P-induced crystal–fluid interaction of a natural paulingite-K have been explored on the basis of in-situ single-crystal and powder X-ray diffraction, and in-situ single-crystal Raman spectroscopy with a diamond anvil cell and a series of diverse pressure-transmitting fluids (i.e., silicone-oil, methanol:ethanol = 4:1, methanol:ethanol:water = 16:3:1). No evidence of any phase transition was observed within the P-range investigated, independent on the used P-fluids. The compressional behavior of paulingite is significantly different in response to the different nature of the P-fluids. A drastically lower compressibility is observed when the zeolite is compressed in methanol:ethanol or, even more noticeably, in methanol:ethanol:water mix. We ascribe this phenomenon to the different crystal–fluid interaction at high pressure: (1) silicone-oil is a “non-penetrating” P-medium, because of its polymeric nature, whereas (2) methanol–ethanol and water are “penetrating” P-fluids. The P-induced penetration processes appear to be completely reversible on the basis of the X-ray diffraction data alone. The Raman spectra collected after the high-pressure experiments show, unambiguously, that a residual fraction of methanol (and/or ethanol and probably even extra H2O) still resides in the zeolitic sub-nanocavities; such molecules are spontaneously released after a few days at atmospheric pressure. The actual compressibility of paulingite-K is that obtained by the compression experiment in silicone-oil, with an isothermal bulk modulus K0 = β0−1 = 18.0(1.1) GPa. Paulingite appears to be one of the softest zeolite ever found.