“Intense Reactivity in Sulfur−Hydrogen Mixtures at High Pressure under X‑ray Irradiation”
- Authors
E.J. Pace, A.L. Coleman, R.J. Husband, H. Hwang, J. Choi, T. Kim, G. Hwang, S.H Chun, D. Nam, S. Kim, O.B. Ball, H.-P. Liermann, M.I. McMahon, Y. Lee, and R.S. McWilliams
- Journal
Journal of Physical Chemistry Letters
Vol.11, pp.1828-1834, 2020.02 - DOI
Abstract
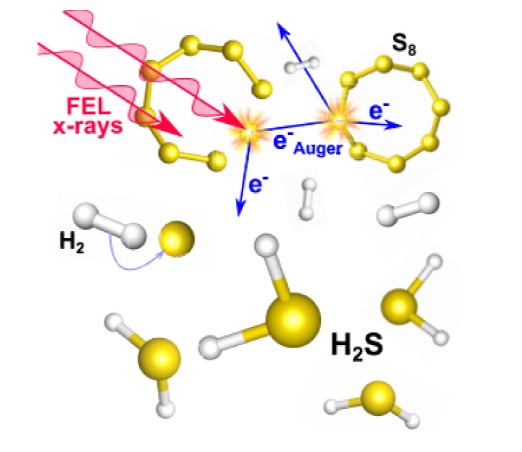
Superconductivity near room temperature in the sulfur–hydrogen system arises from a sequence of reactions at high pressures, with X-ray diffraction experiments playing a central role in understanding these chemical–structural transformations and the corresponding S:H stoichiometry. Here we document X-ray irradiation acting as both a probe and as a driver of chemical reaction in this dense hydride system. We observe a reaction between molecular hydrogen (H2) and elemental sulfur (S8) under high pressure, induced directly by X-ray illumination, at photon energies of 12 keV using a free electron laser. The rapid synthesis of hydrogen sulfide (H2S) at 0.3 GPa was confirmed by optical observations, spectroscopic measurements, and microstructural changes detected by X-ray diffraction. These results document X-ray induced chemical synthesis of superconductor-forming dense hydrides, revealing an alternative production strategy and confirming the disruptive nature of X-ray exposure in studies on high-pressure hydrogen chalcogenides, from water to high-temperature superconductors.